Pre-Topic Activities:
1 - Create a Title Page for "Properties of Substances"
2 - Create a KWH for "Properties of substances" (red the green box above to spark ideas)
- What you Know
- What you Want to know
- How you will learn it
2 - Create a KWH for "Properties of substances" (red the green box above to spark ideas)
- What you Know
- What you Want to know
- How you will learn it
Learning Intention- Know solids, liquids and gases.
- Understand what makes these 3 states different. |
Success Criteria- I can use a model to understand how liquids expand and contract.
|
|
Student Activities - Part 1
1 - Put the heading Solids, Liquids, Gases - Physical Properties" 2 - Complete the "Science 4 fun activity" ----> Follow the instructions carefully. Do not be silly with the foam! 3 - When you finish: Write a Purpose, Apparatus list, Risk Assessment and Procedure for the activity. 4 - Create 3 mind maps on your page: i) Solids ii) Liquids iii) Gases Around each mind map, name as many objects as you can that belong in that heading (example - water is a liquid.) Scroll down for more activities! |
|
Student Activities Part 2
Read the information on the right to complete the following activities: 1 - Define Physical Property 2 - Record the 7 "Most useful Properties Of a Substance" in your book. 3 - What is meant by the words "States of Matter"? 4 - Define the word Compressed. 5 - Define the word Incompressible. 6 - Write 3 facts about: a) A solid b) A liquid c) A Gas 7 - Is there only 3 states of matter? Explain your answer. |
Student Activities Part 3
- Watch the videos below "with headphones if you have them"
- Record 10-15 dotpoints under the heading "Solids, Liquids and Gases Video"
|
|
|
Test Yourself
Learning Intention
|
Success Criteria
|
|
STUDENT ACTIVITIES
- Put the heading "Chemical Properties of Substances" - Define Chemical Properties. - Record the 4 dot points on the right. - Consider the following: a) why is steel used in a skyscraper? b) why is air used in a jumping castle? c) why are tyres made of rubber? d) why is sun screen a liquid? - Complete the "mass of gas" prac below with a full prac write up. |
Copy the Diagram Below
Extension
Learning IntentionI know how to read and interpret secondary sources.
I know what the properties are of solids, liquids and gases. |
Success CriteriaI can present my knowledge in a poster.
I can complete the quiz at the end of the lesson. |
|
STUDENT ACTIVITIES
1 - Define the Particle Model 2 - What does a particle with lots of energy do? 3 - What does a particle with little energy do? 4 - What happens when you add heat to particles? 5 - What happens when you remove heat from particles? 6 - State the 4 things that the particle model assumes: 7 You are to use the video above, and the information below to identify the properties of Solids, Liquids and Gases in the form of A POSTER. - Use a full page for each state of matter - Create a list of features for each state of matter. - Draw a picture of the particles in the state of matter - Provide a lift of examples of that state of matter. 8 - Complete the quiz |
SOLIDS
LIQUIDS
GASES
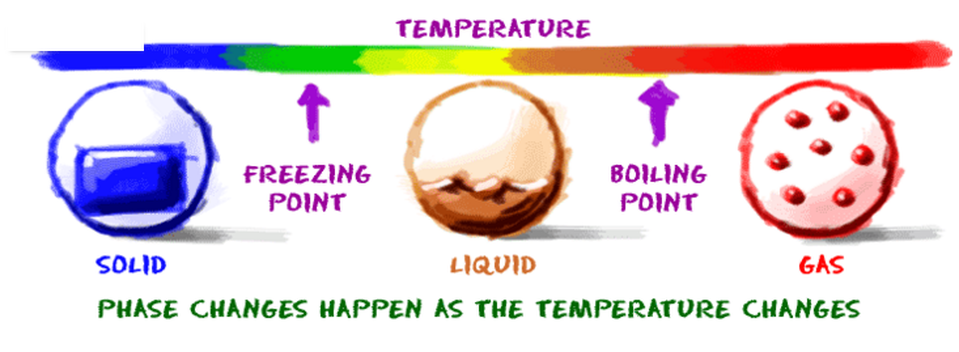
Learning Intention- To know how water changes state
- To understand the temperature changes involved in the changes in state of water - To graph the temperature changes as water changes from liquid to gas |
Success CriteriaUse the SST to correctly describe the transformation of substances between solids, liquids and gases.
|
SST (Subject specific terminology) |
Boiling, boiling point, condensation, deposition, evaporations, vaporisation, freezing, solidification, freezing point, melting, melting point, steam, sublimation
|
Student Activities Part 1:
Use the internet to define each of the subject specific terms above.
Draw the diagram below into your workbook.
Watch the video below and complete the interactive activity.
Use the internet to define each of the subject specific terms above.
Draw the diagram below into your workbook.
Watch the video below and complete the interactive activity.
|
|
|
Interactive Activity
Student Activities Part 2:
Use the text below complete the test yourself section.
Use the text below complete the test yourself section.
|
Creating a Solid
All matter can move from one state to another. It may require extreme temperatures or extreme pressures, but it can be done. Sometimes a substance doesn't want to change states. You have to use all of your tricks when that happens. To create a solid, you have to decrease the temperature by a huge amount and then add pressure. Some of you know about liquid nitrogen (N2). It is nitrogen from the atmosphere in a liquid form and it has to be super cold to stay a liquid. What if you wanted to turn it into a solid but couldn't make it cold enough to solidify? You could increase the pressure in a sealed chamber. Eventually you would reach a point where the liquid became a solid. |
|
Creating a Gas
When you are a liquid and want to become a gas, you need to find a lot of energy. Once you can direct that energy into your molecules, they will start to vibrate. If they vibrate enough, they can escape the limitations of the liquid environment and become a gas. When you reach your boiling point, the molecules in your system have enough energy to become a gas. |
|
Creating a Liquid
The reverse is true if you are a gas. You need to lose some energy from your very excited gas atoms. The easy answer is to lower the surrounding temperature. When the temperature drops, energy will be transferred out of your gas atoms into the colder environment. When you reach the temperature of the condensation point, you become a liquid. If you were steam over a boiling pot of water and you hit a wall, the wall would be so cool that you would quickly become a liquid. The wall absorbed some of your extra energy. |
|
Sublimation and Deposition: Skipping the liquid phase.
Sublimation is the process by which a solid transforms into a gas without passing through the liquid stage. Deposition occurs when a gas becomes a solid without going through the liquid state of matter. Those of you who live near the equator may not have seen it, but closer to the poles we see frost on winter mornings. Those little frost crystals on plants build up when water vapor from the air becomes a solid on the leaves of plants. |
|
|
|
|
Test Yourself
Complete each of these activities in your workbook. NOTE for A, redraw the diagram into your workbook.
Learning IntentionStudents to explain expansion and contraction
|
Success CriteriaWrite a structured paragraph that explains what happens in the video
Successfully explain what happens in each prac |
Student Activities:
Heading in book: Expansion and contraction.
Write the text below into your workbook.
Use it to define the terms expansion and contraction.
Heading in book: Expansion and contraction.
Write the text below into your workbook.
Use it to define the terms expansion and contraction.
Student Activities Continued:
Watch the video below and use it to explain the following:
1 .What is happening to particles when an object expands?
2. What is happening to particles when an object contracts?
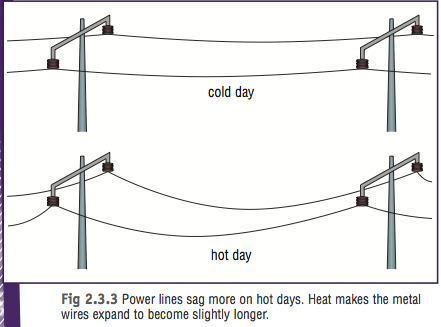
3. Relate it to the image of the power lines below. Use the principle of expansion and contraction to explain whats going on in the image.
Watch the video below and use it to explain the following:
1 .What is happening to particles when an object expands?
2. What is happening to particles when an object contracts?
3. Relate it to the image of the power lines below. Use the principle of expansion and contraction to explain whats going on in the image.
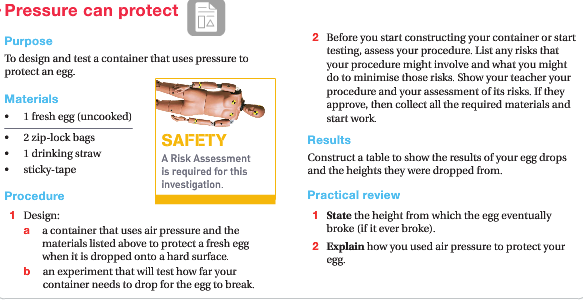
Practical Investigation
Have a go at completing both of these practical activities and complete a Risk assessment for both activities.
NOTE: Remember when writing a risk assessment you need to
Have a go at completing both of these practical activities and complete a Risk assessment for both activities.
NOTE: Remember when writing a risk assessment you need to
- Identify the rich and hazard that causes it
- List some strategies to avoid this or to treat any injury that occurs.
Learning Intention
|
Success Criteria
|
Student Activities Part 1:
Heading in book Density.
Have a look at the image below and complete the activities in your workbook.:
Heading in book Density.
Have a look at the image below and complete the activities in your workbook.:
- Use it to define the term density.
- What can you say about the density of solids in comparison to liquids and gasses and why?
- Have a look at the two beakers of liquid. In full sentences identify which one is more dense and explain why?
|
Student activities continued:
Have a look at the image over there -----------------------------> Read the text below it and use it to answer the following question.
Thinking question: Does 1kg of feathers weigh the same as 1kg of rocks? If they were in a wheelbarrow would they look to be the same size? Put a sub heading in your book that says calculating Density. Copy the information below into your workbook. |
|
Student Activities Continued
Watch the video over there ----------> and use it to answer the questions below into your workbook
Complete the test yourself section below in your workbook. |
|
|
Test yourself
Complete each of the questions in your workbook: NOTE you will probably need calculator. (If you're struggling scroll up to the calculating density box and use that formula to help you. • A rock has a mass of 150g and a volume of 30cm3 Calculate its Density • A match has a mass of 3g and a volume of 12cm3 Calculate its Density • A train has a mass of 152701g and a volume of 1309cm3 Calculate its Density |
|
|
Student Activities:
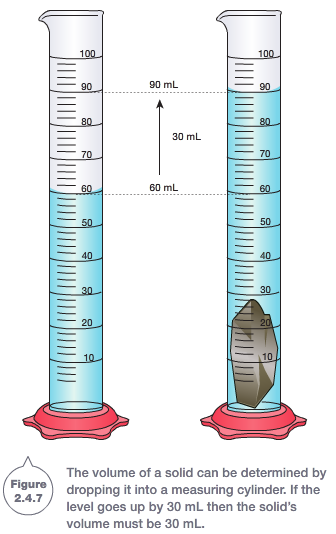
If we don't know the volume of an object because it has an irregular shape an easy way to find it is by placing it in water. The amount of millilitres that the water increases from its original amount shows the volume of the irregular object.
|
Videos you need to watch about density.
|
|
|
| hot_air_balloons_internet_research.docx | |
| File Size: | 8 kb |
| File Type: | docx |
Learning Intention |
Success Criteria |